Bile is a bitter, yellow to greenish fluid composed of glycine or taurine
conjugated bile salts, cholesterol, phospholipid, bilirubin diglucuronide, and
electrolytes. It is secreted by the liver and delivered to the duodenum to aid
the process of digestion and fat absorption by emulsification of fat products in
the upper small intestine. They play role dissolving cholesterol and accretes
into lumps in the gall bladder, forming gallstones. Bile's bicarbonate
constituents serves for alkalinizing the intestinal contents. Bile is
responsible for as the route of excretion for hemoglobin breakdown products
(bilirubin). Excretion of bile salts by liver cells and secretion of bicarbonate
rich fluid by ductular cells in response to secretin are the major factors which
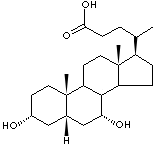
normally determine the volume of secretion. Bile acids are liver-generated
steroid carboxylic acids. The primary bile acids (cholic and chenodeoxycholic
acid) are conjugated with either glycine or taurine in the form of taurocholic
acid and glycocholic acid. The secondary bile acids (deoxycholic, lithocholic,
and ursodeoxycholic acid) are formed from the primary bile acids by the action
of intestinal bacteria. They are soluble in alcohol and acetic acid. The
lithocolyl conjugates are relatively insoluble; excreted mostly in the form of
sulfate esters like sulfolithocholylglycine. Most of the bile acids are
reabsorbed and returned to the liver via enterohepatic circulation, where, after
free acids are reconjugated, they are again excreted.
- Cholic Acid
- Methyl Cholate
- Deoxycholic
Acid
- Dehydrocholic Acid
- Methyl Deoxycholate
- Chenodeoxycholic Acid
- Methyl
Chenodeoxycholate
- Sodium Glycocholate
- Glycodeoxycholic Acid
- Sodium
Glycodeoxycholate
- Sodium Taurocholate
- Sodium Taurodeoxycholate
- Ursodeoxycholic acid
Commecial bile acids' application fields include:
- Experimental basis of
cathartics, cholagogues
- Preventing
and dissolving gallstones.
- As an
intermediate for the production of corticosteroid structure drugs to treat
various disorders such as pain or inflammation, brain tumors, skin diseases, and
allergic reactions.
- As
detergents to emulsify
biological fats.
- Vaccine delivery system
|